What does glycoside mean?
Definitions for glycoside
ˈglaɪ kəˌsaɪdgly·co·side
This dictionary definitions page includes all the possible meanings, example usage and translations of the word glycoside.
Princeton's WordNet
glycosidenoun
a group of compounds derived from monosaccharides
Wiktionary
glycosidenoun
A molecule in which a sugar group (the glycone) is bound to a non-sugar group (the corresponding aglycone) by a nitrogen or oxygen atom. Glycosides yield a sugar after undergoing hydrolysis.
Etymology: From glyco-, + -ide (similar to glucoside), 1925–1930.
Wikipedia
Glycoside
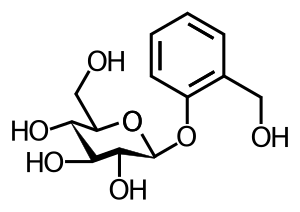
In chemistry, a glycoside is a molecule in which a sugar is bound to another functional group via a glycosidic bond. Glycosides play numerous important roles in living organisms. Many plants store chemicals in the form of inactive glycosides. These can be activated by enzyme hydrolysis, which causes the sugar part to be broken off, making the chemical available for use. Many such plant glycosides are used as medications. Several species of Heliconius butterfly are capable of incorporating these plant compounds as a form of chemical defense against predators. In animals and humans, poisons are often bound to sugar molecules as part of their elimination from the body. In formal terms, a glycoside is any molecule in which a sugar group is bonded through its anomeric carbon to another group via a glycosidic bond. Glycosides can be linked by an O- (an O-glycoside), N- (a glycosylamine), S-(a thioglycoside), or C- (a C-glycoside) glycosidic bond. According to the IUPAC, the name "C-glycoside" is a misnomer; the preferred term is "C-glycosyl compound". The given definition is the one used by IUPAC, which recommends the Haworth projection to correctly assign stereochemical configurations.Many authors require in addition that the sugar be bonded to a non-sugar for the molecule to qualify as a glycoside, thus excluding polysaccharides. The sugar group is then known as the glycone and the non-sugar group as the aglycone or genin part of the glycoside. The glycone can consist of a single sugar group (monosaccharide), two sugar groups (disaccharide), or several sugar groups (oligosaccharide). The first glycoside ever identified was amygdalin, by the French chemists Pierre Robiquet and Antoine Boutron-Charlard, in 1830.
Wikidata
Glycoside
In chemistry, a glycoside is a molecule in which a sugar is bound to a non-carbohydrate moiety, usually a small organic molecule. Glycosides play numerous important roles in living organisms. Many plants store chemicals in the form of inactive glycosides. These can be activated by enzyme hydrolysis, which causes the sugar part to be broken off, making the chemical available for use. Many such plant glycosides are used as medications. In animals and humans, poisons are often bound to sugar molecules as part of their elimination from the body. In formal terms, a glycoside is any molecule in which a sugar group is bonded through its anomeric carbon to another group via a glycosidic bond. Glycosides can be linked by an O-, N-, S-, or C- glycosidic bond. The given definition is the one used by IUPAC, which recommends the Haworth projection to correctly assign stereochemical configurations. Many authors require in addition that the sugar be bonded to a non-sugar for the molecule to qualify as a glycoside, thus excluding polysaccharides. The sugar group is then known as the glycone and the non-sugar group as the aglycone or genin part of the glycoside. The glycone can consist of a single sugar group or several sugar groups.
Matched Categories
Numerology
Chaldean Numerology
The numerical value of glycoside in Chaldean Numerology is: 3
Pythagorean Numerology
The numerical value of glycoside in Pythagorean Numerology is: 9
Popularity rank by frequency of use
Translation
Find a translation for the glycoside definition in other languages:
Select another language:
- - Select -
- 简体中文 (Chinese - Simplified)
- 繁體中文 (Chinese - Traditional)
- Español (Spanish)
- Esperanto (Esperanto)
- 日本語 (Japanese)
- Português (Portuguese)
- Deutsch (German)
- العربية (Arabic)
- Français (French)
- Русский (Russian)
- ಕನ್ನಡ (Kannada)
- 한국어 (Korean)
- עברית (Hebrew)
- Gaeilge (Irish)
- Українська (Ukrainian)
- اردو (Urdu)
- Magyar (Hungarian)
- मानक हिन्दी (Hindi)
- Indonesia (Indonesian)
- Italiano (Italian)
- தமிழ் (Tamil)
- Türkçe (Turkish)
- తెలుగు (Telugu)
- ภาษาไทย (Thai)
- Tiếng Việt (Vietnamese)
- Čeština (Czech)
- Polski (Polish)
- Bahasa Indonesia (Indonesian)
- Românește (Romanian)
- Nederlands (Dutch)
- Ελληνικά (Greek)
- Latinum (Latin)
- Svenska (Swedish)
- Dansk (Danish)
- Suomi (Finnish)
- فارسی (Persian)
- ייִדיש (Yiddish)
- հայերեն (Armenian)
- Norsk (Norwegian)
- English (English)
Word of the Day
Would you like us to send you a FREE new word definition delivered to your inbox daily?
Citation
Use the citation below to add this definition to your bibliography:
Style:MLAChicagoAPA
"glycoside." Definitions.net. STANDS4 LLC, 2024. Web. 6 May 2024. <https://www.definitions.net/definition/glycoside>.


Discuss these glycoside definitions with the community:
Report Comment
We're doing our best to make sure our content is useful, accurate and safe.
If by any chance you spot an inappropriate comment while navigating through our website please use this form to let us know, and we'll take care of it shortly.
Attachment
You need to be logged in to favorite.
Log In