What does triple point mean?
Definitions for triple point
triple point
This dictionary definitions page includes all the possible meanings, example usage and translations of the word triple point.
Wiktionary
triple pointnoun
The unique temperature and pressure at which the solid, liquid and gas phases of a substance are all in equilibrium.
Wikipedia
Triple point
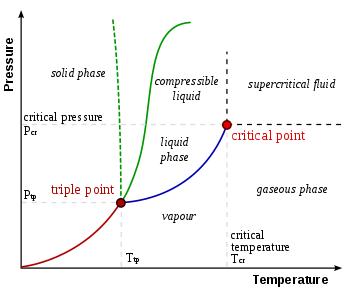
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium. It is that temperature and pressure at which the sublimation curve, fusion curve and the vaporisation curve meet. For example, the triple point of mercury occurs at a temperature of −38.8 °C (−37.8 °F) and a pressure of 0.165 mPa. In addition to the triple point for solid, liquid, and gas phases, a triple point may involve more than one solid phase, for substances with multiple polymorphs. Helium-4 is a special case that presents a triple point involving two different fluid phases (lambda point).The triple point of water was used to define the kelvin, the base unit of thermodynamic temperature in the International System of Units (SI). The value of the triple point of water was fixed by definition, rather than measured, but that changed with the 2019 redefinition of SI base units. The triple points of several substances are used to define points in the ITS-90 international temperature scale, ranging from the triple point of hydrogen (13.8033 K) to the triple point of water (273.16 K, 0.01 °C, or 32.018 °F). The term "triple point" was coined in 1873 by James Thomson, brother of Lord Kelvin.
Wikidata
Triple point
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases of that substance coexist in thermodynamic equilibrium. For example, the triple point of mercury occurs at a temperature of −38.8344 °C and a pressure of 0.2 mPa. In addition to the triple point between solid, liquid, and gas, there can be triple points involving more than one solid phase, for substances with multiple polymorphs. Helium-4 is a special case that presents a triple point involving two different fluid phases. In general, for a system with p possible phases, there are triple points. The triple point of water is used to define the kelvin, the SI base unit of thermodynamic temperature. The number given for the temperature of the triple point of water is an exact definition rather than a measured quantity. The triple points of several substances are used to define points in the ITS-90 international temperature scale, ranging from the triple point of hydrogen to the triple point of water.
Numerology
Chaldean Numerology
The numerical value of triple point in Chaldean Numerology is: 3
Pythagorean Numerology
The numerical value of triple point in Pythagorean Numerology is: 1
Translation
Find a translation for the triple point definition in other languages:
Select another language:
- - Select -
- 简体中文 (Chinese - Simplified)
- 繁體中文 (Chinese - Traditional)
- Español (Spanish)
- Esperanto (Esperanto)
- 日本語 (Japanese)
- Português (Portuguese)
- Deutsch (German)
- العربية (Arabic)
- Français (French)
- Русский (Russian)
- ಕನ್ನಡ (Kannada)
- 한국어 (Korean)
- עברית (Hebrew)
- Gaeilge (Irish)
- Українська (Ukrainian)
- اردو (Urdu)
- Magyar (Hungarian)
- मानक हिन्दी (Hindi)
- Indonesia (Indonesian)
- Italiano (Italian)
- தமிழ் (Tamil)
- Türkçe (Turkish)
- తెలుగు (Telugu)
- ภาษาไทย (Thai)
- Tiếng Việt (Vietnamese)
- Čeština (Czech)
- Polski (Polish)
- Bahasa Indonesia (Indonesian)
- Românește (Romanian)
- Nederlands (Dutch)
- Ελληνικά (Greek)
- Latinum (Latin)
- Svenska (Swedish)
- Dansk (Danish)
- Suomi (Finnish)
- فارسی (Persian)
- ייִדיש (Yiddish)
- հայերեն (Armenian)
- Norsk (Norwegian)
- English (English)
Word of the Day
Would you like us to send you a FREE new word definition delivered to your inbox daily?
Citation
Use the citation below to add this definition to your bibliography:
Style:MLAChicagoAPA
"triple point." Definitions.net. STANDS4 LLC, 2024. Web. 28 Apr. 2024. <https://www.definitions.net/definition/triple+point>.


Discuss these triple point definitions with the community:
Report Comment
We're doing our best to make sure our content is useful, accurate and safe.
If by any chance you spot an inappropriate comment while navigating through our website please use this form to let us know, and we'll take care of it shortly.
Attachment
You need to be logged in to favorite.
Log In